Lauritzen Lab
Research in this laboratory concentrates on the neural and astrocytic mechanisms of local vascular regulation. Our basic research circles around blood flow control and blood-brain barrier properties under normal physiological conditions.

The Lauritzen Laboratory has pioneered research in cerebrovascular biology for over four decades. The laboratory has contributed to our understanding of neurovascular function, including the blood-brain barrier (BBB) and the role of cortical spreading depolarisation/depression (CSD) in migraine and acute brain injury. The first major contribution was the early work on migraine and CSD as a mechanism of this neurological disorder, and later on the role of CSDs in acute neurological injury. A second major focus of the laboratory's research programme has been the mechanistic understanding of neurovascular coupling, i.e. the regulation of cerebral blood flow (CBF) by neural activity and the generation of functional magnetic resonance imaging (fMRI) signals in health, aging and disease. The lab has developed advanced new imaging and image analysis tools for multimodal measurements of brain activity to identify the cellular and molecular origins of neurovascular coupling. A third and more recent focus is the exploration of the BBB by multiphoton microscopy and the quantification of permeability properties along para- and transcellular pathways.
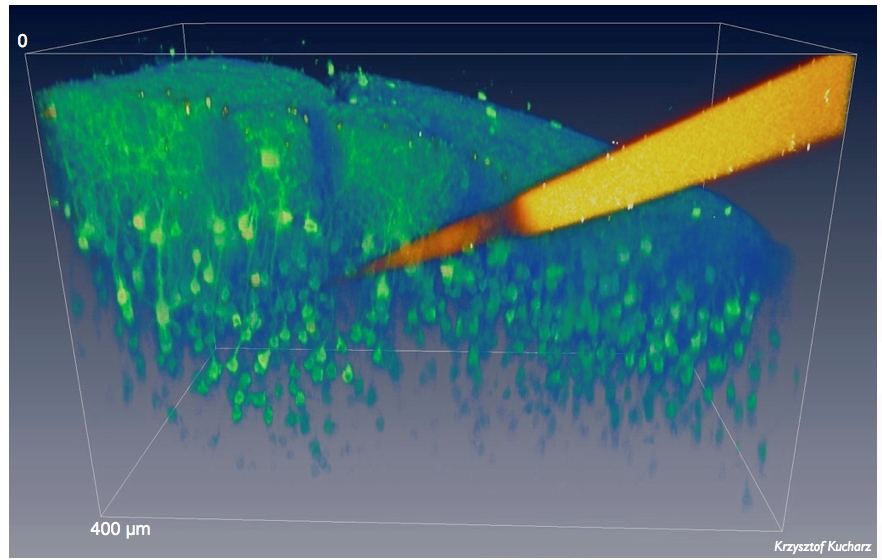
However, drug delivery to the brain is a major challenge due to permeability of the blood-brain barrier (BBB). Emerging therapies for Alzheimer's disease (AD) are based on monoclonal antibodies targeting the amyloid beta protein, but only about 0.1%-0.2% of the administered antibody reaches the brain because the antibodies do not cross the BBB effectively. If macromolecules do enter the brain at all, it is via the choroid plexus. This is ineffective for the treatment of brain disease because only a small amount of antibody is secreted from the plexus into the cerebrospinal fluid, which flows through the ventricles and cisterns to the subarachnoid space. Low brain uptake means that high doses of antibodies must be used to treat Alzheimer's disease, sometimes with severe side effects that can outweigh the benefits of available therapies. We are now using small antibody fragments that can be coupled to clinically approved therapeutic antibodies to target surface proteins on brain endothelial cells. Binding and activation of transport mechanisms on the endothelial surface results in high brain uptake at much lower therapeutic doses. Using two-photon microscopy in awake mice, we are developing this strategy, describing the mechanisms of antibody construct transport across the BBB, biomolecule trafficking in the extracellular space of the brain, and antibody interactions with therapeutic targets in clinically relevant models of Alzheimer's disease and, in a recently funded project, Parkinson's disease.
In summary, the laboratory operates at the interface between neuroscience and clinical neurology, bridging the worlds of biology and technology, advancing two-photon microscopy imaging and applying novel methods to address key neuroscience questions relevant to neurovascular physiology, regulation of blood flow and metabolism, and drug delivery in brain ageing and neurodegenerative diseases.
- Cai C, Zambach S, Grubb S, Tao L, He C, Lind BL, Thomsen KJ, Zhang X, Hald BO, Nielsen RM, Kim K, Devor A, Lønstrup M, Lauritzen MJ. Impaired dynamics of precapillary sphincters and pericytes at first-order capillaries predict reduced neurovascular function in the aging mouse brain. Nature Aging 2023, DOI
1038/s43587-022-00354-1 - Kucharz K, Kristensen K, Johnsen KB, Lund MA, Lønstrup M, Moos T, Andresen TL, Lauritzen M. Post-capillary venules are the key locus for transcytosis-mediated brain delivery of therapeutic Nat Comm 2021 Jul 5; 12(1):4121. doi: 10.1038/s41467-021-24323-1.
- Zambach SA, Cai C, Helms HC, Hald BO, Dongd Y, Fordsmann JC, Nielsen RN, Hud J, Lønstrup M, Brodin B, Lauritzen M. Precapillary sphincters and pericytes at first-order capillaries as key regulators for brain capillary perfusion. Proc Natl Acad Sci U S A 2021, DOI number 10.1073/pnas.2023749118.
- Grubb S, Cai C, Hald BO, Khennouf L, Fordsmann J, Murmu R, Jensen AKG, Zambach S, Lauritzen M. Precapillary sphincters maintain perfusion in the cerebral cortex. Nat Commun. 2020 Jan 20; 11(1):395. doi: 10.1038/s41467-020-14330-z. PMID: 31959752.
- Cai C, Fordsmann J, Jensen SH, Hald BO, Gesslein B, Lønstrup M, Brodin B, Lauritzen M. Conducted vascular responses in brain capillaries by synaptic activity and ATP in mouse cerebral cortex. Proc Natl Acad Sci U S A. 2018 Jun 19; 115(25):E5796-E5804. oi:.1073/pnas.1707702115. PMID: 29866853.
- Kutuzov N, Flyvbjerg H, Lauritzen M. Contributions of the glycocalyx, endothelium, and extravascular compartment to the blood-brain barrier. Proc Natl Acad Sci U S A. 2018 Oct 2; 115(40):E9429-E9438. doi: 10.1073/pnas.1802155115.
- Khennouf L, Gesslein B, Brazhe A, Octeau JC, Kutuzov N, Khakh BS, Lauritzen M. Active role of capillary pericytes during stimulation-induced activity and spreading depolarization. Brain 2018, July, 141 (7); 2032–2046.
- Ayata C, Lauritzen M: Spreading Depression, Spreading Depolarizations, and the Cerebral Vasculature. Physiol Rev 2015; 95:953-993.
- Hall CN, Reynell C, Gesslein B, Hamilton NB, Mishra A, Sutherland BA, O'Farrell FM, Buchan AM, Lauritzen M**, Attwell D**: Capillary pericytes regulate cerebral blood flow in health and disease. Nature 2014; 508:55-60. **Co-corresponding senior authors.
- Lind BL, Brazhe AR, Jessen SB, Tan FC, Lauritzen M: Rapid stimulus-evoked astrocyte Ca2+ elevations and hemodynamic responses in mouse somatosensory cortex in vivo. Proc Natl Acad Sci U S A 2013; 110:E4678-E4687.
Wikipedia
POSITION TITLE
Professor of Translational Neurobiology, Department of Neuroscience, University of Copenhagen, Copenhagen, Denmark.
Professor emeritus of Clinical Neurophysiology, University of Copenhagen, Rigshospitalet (National University Hospital), Copenhagen, Denmark
EDUCATION/TRAINING
|
INSTITUTION AND LOCATION |
DEGREE |
YEAR(s) |
FIELD OF STUDY |
|
University of Copenhagen |
MD |
1978 |
Medicine |
|
University of Copenhagen |
Research fellow |
1978-1985 |
Neurology and neurobiology |
|
New York University Medical Center |
Fulbright Fellow |
1986 |
Neurobiology |
|
University of Copenhagen and Copenhagen University Hospitals |
Internship and residencies |
1987-1994 |
Neurology, Neurosurgery, Clinical Neurophysiology |
|
University of Copenhagen |
Dr.med.sci |
1988 |
Neurobiology |
A. POSITIONS AND HONORS
Appointments and other professional activities
1994 - 2024 Head and Consultant, Department of Clinical Neurophysiology, Copenhagen University Hospital in Glostrup and Rigshospitalet (Danish National Hospital), DK
1998 - 2022 Professor of Clinical Neurophysiology, University of Copenhagen, DK
2007 - present Professor of Translational Neurobiology, University of Copenhagen, DK
2000 - 2018 Director, International Society for Cerebral Blood Flow & Metabolism,
2004 & 2006 Chair: Gordon Research Conference on ‘Brain Energy Metabolism and Blood Flow’.
2004 Visiting Professor, Humboldt University, Germany
Commissions of trust
2016 – 2024 International Scientific Advisory Board Member for IBS Center for Neuroscience Imaging Research, Sungkyunkwan University, South Korea
2009 – 2014 Member of Transatlantic Network of Excellence funded by Foundation Leducq
2009 – 2022 International Scientific Advisory Board Member Max-Planck Institute for Cognitive Neuroscience, Leipzig
2019 Founding Director of
2008 – 2014 International Scientific Advisory Board Member, Max-Planck Institute for Neurology, Cologne, Germany
2009 – 2018 Editor-in-chief, Journal of Cerebral Blood Flow & Metabolism
2018 - Consulting editor, Journal of Cerebral Blood Flow & Metabolism
1998 – Review Board for grant applications numerous times for many foundations, including: Bi-national Science Foundation (USA – Israel), Third World Academy of Science; Grant Academy of Czech Republic; The Migraine Trust, UK; Welcome Foundation, UK; Suisse Research Grant Agency; Medical Research Council, UK; Royal Society, UK; German Research Foundation (Deutsche Forschungsgemeinschaft); Canadian, Butch and Belgian funding agencies.
2000 – 2008 Editorial Board, Journal of Cerebral Blood Flow & Metabolism
2008 – 2010 Editorial Board, Neuroimage
2000 – 2005 Editorial Board, Clinical Neurophysiology
Honors and awards
1992 Arnold Friedman Distinguished Clinician/Researcher Award (American Headache Association)
2004 Humboldt Research Award in Medicine (Alexander von Humboldt Stiftung, Germany).
2004 Elected Fellow of the Humboldt Society
2016 Niels A. Lassen Prize
2017 Member of Academia Europaea (European Academy of Science)
2022 Honorary member of Society of Clinical Neurophysiology
2024 Elected Fellow of the Academy of the International Union of Physiological Sciences
B. RESEARCH MANAGEMENT EXPERIENCE
2014 – 2021 Director of the Lundbeck Foundation Research Initiative on Brain Barriers and Drug Delivery, University of Copenhagen
2025 - 2029 Co-Director of the Lundbeck Foundation Research Initiative on NanoPANS: functionalized nanocarriers sending anti-PD drugs to central nerve systems.
2009 – 2021 Member of Board of Directors, Center for Healthy Aging, University of Copenhagen
2007 – 2022 Member of Board of Directors, Committee for Neurology and Clinical Neurophysiology
Copenhagen, Capital region of Denmark
1998 – Member/Chair for Committees assessing numerous full professorships at University of Copenhagen
1998 – Member of Committees or panels recommending professor- or directorships at e.g. Baylor College of Medicine, Houston Texas; University of Rochester, Rochester, NY; University of Maryland, MD; Harvard University, Massachusetts; UCSD, California; Max-Planck Society; University of Calgary, Canada.
2010 – 2016 Member of the Research Strategy Faculty Committee, University of Copenhagen
2003 – 2007 Chairman of Strategic Brain Research Program at University of Copenhagen
2003 – Founding member of the steering committee for COSBID, a large multi-centre study of the importance of brain injury depolarization for the outcome of patients with trauma, haemorrhage and stroke (www.cosbid.org)
1998 – Examiner of PhD and dr.med.sci-theses in Denmark, France, Sweden, Norway.
C. RESEARCH SUPPORT
2014-2021 Cell-based control of brain energy supply and use and changes with age’, NORDEA Foundation (2,400,000 Euros)
2014-2021 Brain Barriers and Drug Delivery, Lundbeck Foundation (3,000,000 Euros)
2021-2024 Brain microvascular dynamics in awake mice in health and post-concussion syndrome, Danish Foundation for Independent Research (830.000 Euro).
2021-2022 Rescuing the blood-brain barrier in Huntington's disease, The Friis Foundation (134.000 Euro).
2019-2023 Cerebral blood flow, brain work and stroke, The NOVO Nordisk Foundation, (335.000 Euro)
2024-2026 Two-photon imaging of antibody-drug shuttles to improve immunotherapies in Alzheimer’s disease, the pharmacokinetics, The Lundbeck Foundation (530.000 Euro)
2024-2026 Targeting beta-amyloid in Alzheimer’s disease by therapeutic antibodies coupled to brain shuttles, the pharmacodynamics, The NOVO Nordisk Foundation (400.000 EURO).
2025-2029 NanoPANS: functionalized nanocarriers sending anti-PD drugs to central nerve system. The Lundbeck Foundation (900.000 Euro)
D. SUPERVISION OF GRADUATE STUDENTS AND POSTDOCTORAL FELLOWS
1994 – I have trained more than 35 PhD students and more than 20 postdocs in my lab, most of whom now have leading positions in hospitals in Copenhagen, at University of Copenhagen or in international pharmaceutical companies. I was a member of the BODs setting up “The Graduate School for Neuroscience” at UCPH in 1996 and have served continuously on its board till 2020. I arrange PhD courses biannually, on “Brain energy metabolism and blood flow” and ‘Experimental deign and data analysis’.
E. TEACHING ACTIVITIES
2019 Director of the CAJAL Advanced Neuroscience Training Program, ‘Brain Homeostasis and Neurovascular Coupling’, Bordeaux, France.
2007 – Teaching basic neuroscience and acting supervisor for medical students' bachelor and master theses in translational neurobiology as part of my job as professor of translational neurobiology at the Faculty of Health and Medical Sciences, University of Copenhagen, Denmark.
1994 – Teaching neurology and clinical neurophysiology and acting supervisor for clinically relevant bachelor and master theses as part of my job as clinical professor at the Faculty of Health and Medical Sciences, University of Copenhagen, Denmark.
1994-2022 Teaching Clinical Neurophysiology as part of my job as clinical professor at the Faculty of Health and Medical Sciences, University of Copenhagen, Denmark to 100 neurologists during their 4-6 months educational stay in my hospital department.
F. MAJOR COLLABORATIONS
National collaboration with scientists at the Center for Healthy Aging, UCPH on the mechanisms of cognitive decline in brain aging including animal experimental and human research. Professors Henrik Larsson, Merete Osler, Erik Lykke Mortensen and Krisztina Bendek, University of Copenhagen on brain and cognitive aging.
Within the framework of the Lundbeck Foundation NANOPans Research Initiative collaborations with Professor Daniel Otzen at Aarhus University, and Professor Nikos Hatzakis at University of Copenhagen. We collaborate with Professor Morten Meldal at the University of Copenhagen on beta-bodies for treatment of Alzheimer’s disease. We build mathematical models of capillary function and superlocalization microscopy with Professor Henrik Flyvbjerg and his group at Danish Technical University.
International collaborations: Professors Ulrich Dirnagl and Jens Dreier, Klinikum Charité, Humboldt University, Berlin on stroke models and brain injury depolarization waves; Professor Pete Tessier at University of Michigan and Professors Stina Syvanen and Dag Sehlin, Uppsala University on monoclonal antibodies for Alzheimer’s disease; Professor Gerald Dienel at Arizona University on metabolic water and brain fluid flow. Professor Anna Devor at Boston University on new developments in two photon microscopy. Professor Cenk Ayata at Harvard Medical school on brain depolarization waves.
G. PUBLICATIONS
Number of peer-reviewed publications
Total 209 peer-reviewed papers and reviews
Total 38 book chapters. Ten editorials. Book chapters and editorials are omitted from publication list.
Top publications from 2013-2025
- Cai C, Zambach S, Grubb S, Tao L, He C, Lind BL, Thomsen KJ, Zhang X, Hald BO, Nielsen RM, Kim K, Devor A, Lønstrup M, Lauritzen MJ. Impaired dynamics of precapillary sphincters and pericytes at first-order capillaries predict reduced neurovascular function in the aging mouse brain. Nature Aging 2023 Feb;3(2):173-184. doi:1038/s43587-022-00354-1.
- Kucharz K, Kristensen K, Johnsen KB, Lund MA, Lønstrup M, Moos T, Andresen TL, Lauritzen M. Post-capillary venules are the key locus for transcytosis-mediated brain delivery of therapeutic Nat Comm 2021 Jul 5; 12(1):4121. doi: 10.1038/s41467-021-24323-1.
- Zambach SA, Cai C, Helms HC, Hald BO, Dongd Y, Fordsmann JC, Nielsen RN, Hud J, Lønstrup M, Brodin B, Lauritzen M. Precapillary sphincters and pericytes at first-order capillaries as key regulators for brain capillary perfusion. Proc Natl Acad Sci U S A 2021, 118(26):e2023749118. doi:1073/pnas.2023749118.
- Grubb S, Cai C, Hald BO, Khennouf L, Fordsmann J, Murmu R, Jensen AKG, Zambach S, Lauritzen M. Precapillary sphincters maintain perfusion in the cerebral cortex. Nat Commun. 2020 Jan 20; 11(1):395. doi:1038/s41467-020-14330-z.
- Cai C, Fordsmann J, Jensen SH, Hald BO, Gesslein B, Lønstrup M, Brodin B, Lauritzen M. Conducted vascular responses in brain capillaries by synaptic activity and ATP in mouse cerebral cortex. Proc Natl Acad Sci U S A. 2018 Jun 19; 115(25):E5796-E5804. doi: 1073/pnas.1707702115.
- Kutuzov N, Flyvbjerg H, Lauritzen M. Contributions of the glycocalyx, endothelium, and extravascular compartment to the blood-brain barrier. Proc Natl Acad Sci U S A. 2018 Oct 2; 115(40):E9429-E9438. doi:1073/pnas.1802155115.
- Khennouf L, Gesslein B, Brazhe A, Octeau JC, Kutuzov N, Khakh BS, Lauritzen M. Active role of capillary pericytes during stimulation-induced activity and spreading depolarization. Brain 2018, July, 141 (7); 2032–2046. doi:1093/brain/awy143.
- Ayata C, Lauritzen M: Spreading Depression, Spreading Depolarizations, and the Cerebral Vasculature. Physiol Rev 2015; 95:953-993. doi:1152/physrev.00027.2014.
- Hall CN, Reynell C, Gesslein B, Hamilton NB, Mishra A, Sutherland BA, O'Farrell FM, Buchan AM, Lauritzen M**, Attwell D**: Capillary pericytes regulate cerebral blood flow in health and disease. Nature 2014; 508:55-60. **Co-corresponding senior authors. doi:1038/nature13165.
- Lind BL, Brazhe AR, Jessen SB, Tan FC, Lauritzen M: Rapid stimulus-evoked astrocyte Ca2+ elevations and hemodynamic responses in mouse somatosensory cortex in vivo. Proc Natl Acad Sci U S A 2013; 110:E4678-E4687. doi:1073/pnas.1310065110.
H. Invited lectures.
48 invited lectures from 2015 to 2024. See enclosure 1 for details
I. Bibliometrics
Google Scholar Citations 26.941, HI: 76
In the Lauritzen lab we perform our studies using a wide range of techniques:
- Calcium imaging
- Two-photon imaging microscopy
- Blood vessel imaging
- In vivo brain studies
- Oxygen measurements
- Electrophysiology
- Laser-Doppler flowmetry
- Laser-speckle imaging
- Whisker stimulation
- Optogenetics
- Virus injections of genetically encoded calcium indicators
Articles in peer-reviewed journals (book chapters and editorials are not in list).
- Lauritzen M, Damgaard JJ, Rubin I, Lauritzen E. A comparison of the properties of renin isolated from pig and rat kidney. Biochem. J., 1976. 155 (2):317-323. doi: 10.1042/bj1550317.
- Rubin I, Lauritzen E, Lauritzen M. Studies on the native forms of renin in the rat kidney. Biochem. Biophys. Acta, 1980. 612:126-136. doi: 10.1016/0005-2744(80)90285-5.
- Lauritzen E, Lauritzen M, Rubin I. Purification of rat renal renin from crude kidney extracts by deaminohexamethylene - Sepharose chromatography. Biochem. Biophys. Res. Comm., 1980. 98 (2):907-914. https://doi.org/10.1016/0006-291X(80)91441-2
- Lauritzen M, Smith T, Fischer-Hansen B, Sparup J, Olesen J. Eaton-Lambert syndrome and malignant thymoma. Neurology, 1980. 30:634-38. doi: 10.1212/wnl.30.6.634.
- Olesen J, Larsen B, Lauritzen M. Focal hyperemia followed by spreading reduction and impaired activation of rCBF in classical migraine. Ann. Neurol. 1981. 344-352. doi: 10.1002/ana.410090406.
- Lauritzen M, Alving J, Paulson OB. Orbital bruits in internal carotid artery occlusion. Clin. Neurol. Neurosurg., 1981. 83:7-10. DOI: 1016/s0303-8467(81)80003-0
- Lauritzen M, Henriksen L, Lassen NA. Regional cerebral blood flow during rest and skilled hand movements by Xenon-133 inhalation and emission computerized tomography. J Cereb Blood Flow Metab, 1981, 1:385-389. doi: 10.1038/jcbfm.1981.42.
- Lauritzen M, Trojaborg W, Olesen J. EEG during attacks of common and classical migraine. Cephalalgia, 1981, 1: 63-66. doi: 10.1111/j.1468-2982.1981.tb00010.x.
- Henriksen L, Paulson OB, Lauritzen M. The effects of sodium nitroprusside on cerebral blood flow and cerebral venous blood gasses. Observations in awake man during and following moderate blood pressure reduction. Eur J Clin Invest, 1982. 12:383-387. doi: 10.1111/j.1365-2362.1982.tb00684.x.
- Lauritzen M, Balslev Jørgensen M, Diemer NH, Gjedde A, Hansen AJ. Persistent oligemia of rat cerebral cortex in wake of spreading depression. Ann Neurol, 1982. 12:469-474. doi: 10.1002/ana.410120510.
- Clifford T, Lauritzen M, Bakke M, Olesen J, Møller E. Electromyography of pericranial muscles during treatment of spontaneous common migraine attacks. Pain, 1982. 14:137-147. doi: 10.1016/0304-3959(82)90094-X
- Tfelt-Hansen P, Jensen K, Vendsborg P, Lauritzen M, Olesen J. Chlormezanone in the treatment of migraine attacks. A double-blind comparison with diazepam and placebo. Cephalalgia, 1982. 2 :205-210. doi: 10.1046/j.1468-2982.1982.0204205.x.
- Olesen J, Lauritzen M, Tfelt-Hansen P, Henriksen L, Larsen B. Spreading cerebral oligemia in classical and normal cerebral blood flow in common migraine. Headache, 1982. 22: 242-248. doi: 10.1111/j.1526-4610.1982.hed2206242.x.
- Lauritzen M, Olsen TS, Lassen NA, Paulson OB. Changes in regional cerebral blood flow during the course of classic migraine attacks. Ann Neurol, 1983. 13:633-641. doi: 10.1002/ana.410130609.
- Lauritzen M, Olsen TS, Lassen NA, Paulson OB. Regulation of regional cerebral blood flow during and between migraine attacks. Ann Neurol 1983. 14:569-572. doi: 10.1002/ana.410140512.
- Hansen BA, Sørensen PS, Lauritzen M, Iversen J, Lauland S, Petrera JE, Nielsen JB, Paulson OB. A case of malignant lymphoma and myasthenia gravis. Scand J Haematol, 1983. 31: 155-160.
DOI: 10.1111/j.1600-0609.1983.tb01524.x
- Lauritzen M, Olesen J. Regional cerebral blood flow during migraine attacks by Xenon-133 inhalation and emission tomography. Brain, 1984. 107:447-461. doi: 10.1093/brain/107.2.447.
- Lauritzen M. Long-lasting reduction of cortical blood flow of the rat brain after spreading depression with preserved autoregulation and impaired CO2 response. J. Cereb Blood Flow Metab, 1984. 4:546-554. doi: 10.1038/jcbfm.1984.79.
- Hansen AJ, Lauritzen M. The role of spreading depression in acute brain disorders. Anais da Academia Brasileira de Ciencias, 1984. 6:457-479. PMID: 6398638
- Shirahata N, Henriksen L, Vorstrup S, Holm S, Lauritzen M, Paulson OB, Lassen NA. Regional cerebral blood flow by Xenon-133 inhalation and emission tomography. Normal values. J Comput Ass Tomog 1985. 9:861-866. doi: 10.1097/00004728-198509000-00004.
- Jensen K, Tfelt-Hansen P, Lauritzen M, Olesen J. Clinical trial of nimodipine for single attacks of classic migraine. Cephalalgia 1985. 5:125-133. doi: 10.1046/j.1468-2982.1985.0503125.x.
- Lauritzen M. EEG-depression/Spreading cortical depression -mulig patofysiologisk model ved migræne. Nord Med 1985. 100:286-288 PMID: 4069989
- Olesen J, Lauritzen M. Migræne og hjernens regionale blodgennemstrømning. Nord Med1985.100:284-285. PMID: 4069988.
- Lauritzen M. Beitrag zur Pathogenese der Störungen in der Hirndurchblutung bei Migräne. Therapie Woche 1985. 35: 3786-3793. Doi not available.
- Olesen J., Vorstrup S, Lauritzen M. rCBF after TIA and during migraine attacks. Cephalalgia, 1985. Suppl 2: 43-46. doi: 10.1177/03331024850050S207.
- Lauritzen M, Diemer NH. Uncoupling of cerebral blood flow and metabolism after single episodes of cortical spreading depression in the rat brain. Brain Res 1986. 370:405-408. doi: 10.1016/0006-8993(86)90504-4.
- Jensen K, Tfelt-Hansen P, Lauritzen M, Olesen J (1986). Classic migraine. A prospective recording of symptoms. Acta Neurol Scand 73:359-362. doi: 10.1111/j.1600-0404.1986.tb03290.x.
- Lauritzen M. Regional cerebral blood flow during cortical spreading depression in rat brain: Increased reactive hyperemia in low-flow states. Acta Neurol Scand 1987. 75:1-8. doi: 10.1111/j.1600-0404.1987.tb07881.x.
- Wahl M, Lauritzen M, Schilling L. Change of cerebrovascular reactivity after cortical spreading depression in cats and rats. Brain Res 1987. 411:72-80. doi: 10.1016/0006-8993(87)90682-2.
- Okada Y, Lauritzen M, Nicholson C. MEG source models and physiology. Phys Med Biol 1987. 32:43-51. doi: 10.1088/0031-9155/32/1/007.
- Okada Y, Lauritzen M, Nicholson C. Magnetic field associated with neural activities in an isolated turtle cerebellum. Brain Res 1987. 412:151-155. doi: 10.1016/0006-8993(87)91451-x.
- Lauritzen M. Cortical spreading depression as a putative migraine mechanism. Trends in Neuroscience 1987. 10:8-13. https://doi.org/10.1016/0166-2236(87)90115-9
- Lauritzen M. Cerebral blood flow in migraine and cortical spreading depression. Thesis. Acta Neurol Scand (Suppl 113), 1987. 76:1-40. PMID: 3324620.
- Okada Y, Lauritzen M, Nicholson C. Magnetic field associated with spreading depression: A model for the detection of migraine. Brain Res, 1988, 442:185-190. doi: 10.1016/0006-8993(88)91450-3.
- Lauritzen M, Rice ME, Okada Y, Nicholson C. Quisqualate, kainate, and NMDA can initiate spreading depression in the turtle cerebellum. Brain Res, 1988, 475:317-327. doi: 10.1016/0006-8993(88)90620-8.
- Wahl M, Lauritzen M, Schilling L. Cerebrovascular Reactivity after Spreading Depression. Prog Appl Microcirc, 1989, 16: 89-96. Doi not available.
- Lauritzen M, Hansen AJ, Kronborg D, Wieloch T. Cortical spreading depression is associated with arachidonic acid accumulation and preservation of energy charge. J Cereb Blood Flow Metab, 1990, 10:115-122. doi: 10.1038/jcbfm.1990.14.
- Zhang E, Hansen AJ, Wieloch T, Lauritzen M. Influence of MK801 on brain extracellular calcium and potassium activities in severe hypoglycemia. J Cereb Blood Flow Metab, 1990, 10:136-139. doi: 10.1038/jcbfm.1990.18.
- Kronborg D, Dalgaard P, Lauritzen M. Ischemia may be the primary cause of neurological deficits in classic migraine. Letter to the Editor, Archs Neurol, 1990, 47:124-125. DOI: 1001/archneur.1990.00530020018005
- Lauritzen M, Liguori R, Trojaborg W. Orthodromic sensory conduction along the ring finger in normal subjects and in patients with a carpal tunnel syndrome. EEG Clin Neurophysiol, 1991, 81:18-23. doi: 10.1016/0168-5597(91)90099-j.
- Dalgaard P, Kronborg D, Lauritzen M. Migraine with aura, cerebral ischemia, spreading depression, and Comptom scatter. Letter to the Editor, Headache, 1991, 31: 49-51. doi: 10.1111/j.1526-4610.1991.hed3101049.x.
- Zhang E, Mikkelsen JD, Fahrenkrug J, Møller M, Kronborg D, Lauritzen M. Prepro-VIP derived peptide sequences in cerebral blood vessels of rats: On the functional anatomy of metabolic autoregulation. J Cereb Blood Flow Metab, 1991, 11:932-938. doi: 10.1038/jcbfm.1991.158.
- Jakobsen JK, Lauritzen MJ, Krarup C. Kronisk inflammatorisk demyeliniserende polyradikuloneuropati. Ugeskr Læger, 1991, 153:2474-2477. PMID: 1926597
- Lauritzen M, Hansen AJ. The effect of glutamate receptor blockade on anoxic depolarization and cortical spreading depression. J Cereb Blood Flow Metab, 1992, 12:223-229. doi: 10.1038/jcbfm.1992.32.
- Colding-Jørgensen E, Lauritzen M, Johnsen NJ, Mikkelsen KB, Særmark K. On the evidence of auditory magnetic fields as an objective measure of tinnitus. EEG Clin Neurophysiol, 1992, 83: 322-327. doi: 10.1016/0013-4694(92)90091-u.
- Lauritzen M. Spreading depression and Migraine. Path Biol, 1992, 40: 332-337. PMID: 1495817. Doi not available.
- Lauritzen M. Cortical spreading depression as a migraine mechanism: Clinical and experimental aspects. Exp Brain Res, 1992, 23: 7-15. ISSN 0014-4819. Doi not available.
- Fabricius M, Lauritzen M. Transient hyperemia succeeds oligemia in wake of cortical spreading depression. Brain Res, 1993, 602: 350-353. doi: 10.1016/0006-8993(93)90701-n.
- Fabricius M, Jensen LH, Lauritzen M. Microdialysis of interstitial amino acids during spreading depression and anoxic depolarization in rat neocortex. Brain Res, 1993, 612: 61-69. doi: 10.1016/0006-8993(93)91644-8.
- Colding-Jørgensen E, Laursen H, Lauritzen M. Focal myositis of the thigh. Report of two cases. Acta Neurol Scand, 1993, 88: 289-292. doi: 10.1111/j.1600-0404.1993.tb04238.x.
- Fabricius M, Lauritzen M. Examination of the role of nitric oxide for the hypercapnic rise of cerebral blood flow in rats. Am J Physiol, 1994, 266: H1457-1464. doi: 10.1152/ajpheart.1994.266.4.H1457.
- Lauritzen M. Pathophysiology of the migraine aura: The spreading depression theory. Brain, 1994, 117: 199-210. doi: 10.1093/brain/117.1.199.
- Akgören N, Fabricius M, Lauritzen M. Importance of nitric oxide for local increases of blood flow in rat cerebellar cortex during electrical stimulation. Proc Nat Acad Sci (U.S.A.), 1994, 91: 5903-5907. doi: 10.1073/pnas.91.13.5903.
- Fabricius M, Akgören N, Lauritzen M. The arginine - nitric oxide pathway and cerebrovascular regulation in cortical spreading depression. Am J Physiol, 1995, 269: H23-H29. doi: 10.1152/ajpheart.1995.269.1.H23.
- Lauritzen M, Fabricius M. Real time laser-Doppler perfusion imaging of cortical spreading depression in rat neocortex. Neuroreport, 1995, 6: 1271-1273. doi: 10.1097/00001756-199506090-00010.
- Fabricius M, Lauritzen M. Laser-Doppler evaluation of rat brain microcirculation: Comparison with the [14C]-iodoantipyrine method suggests discordance during cerebral blood flow increases. J Cereb Blood Flow Metab, 1996, 16:156-161. doi: 10.1097/00004647-199601000-00018.
- Akgören N, Dalgaard P, Lauritzen M. Cerebral blood flow increases evoked by electrical stimulation of rat cerebellar cortex: relation to excitatory synaptic activity and nitric oxide synthesis. Brain Res, 1996, 710:204-214. doi: 10.1016/0006-8993(95)01354-7.
- Colding-Jørgensen E, Sørensen SE, Hasholt L, Lauritzen M. Electrophysiological findings in a Danish family with Machado-Joseph disease. Muscle & Nerve, 1996, 19:743-750. doi: 10.1002/(SICI)1097-4598(199606)19:6<743::AID-MUS9>3.0.CO;2-A.
- Lauritzen M. Pathophysiology of the migraine aura. Invited review. Science & Medicine, 1996, 3: 32-41. Doi not available.
- Fabricius M, Rubin I, Bundgaard M, Lauritzen M. Nitric oxide synthase activity in brain and endothelium: relation to hypercapnic rise of cerebral blood flow in rats. Am J Physiol, 1996, 40: H2035-H2044. doi: 10.1152/ajpheart.1996.271.5.H2035.
- Andersen B, Lauritzen M. 'En vissen arm'. Multifokal motorisk neuropati og differentialdiagnostiske overvejelser. Ugeskrift for Læger, 1996, 31: 4390-44395. PMID: 8759995
- Barfod C, Akgören N, Fabricius M, Dirnagl U, Lauritzen M. Laser-Doppler measurements of concentration and velocity of moving blood cells in rat cerebral circulation. Acta Physiol Scand, 1997, 160:123-132. doi: 10.1046/j.1365-201X.1997.00130.x.
- Fabricius M, Akgören N, Dirnagl U, Lauritzen M. Laminar analysis of cerebral blood flow in cortex in rats by laser-Doppler flowmetry. A pilot study. J Cereb Blood Flow Metab, 1997, 17:1326-1336. doi: 10.1097/00004647-199712000-00008.
- Akgören N, Mathiesen C, Rubin I, Lauritzen M. Laminar analysis of activity-dependent increases of cerebral blood flow in rat cerebellar cortex: dependence on synaptic strength. Am J Physiol, 1997, 273:H1166-H1176. doi: 10.1152/ajpheart.1997.273.3.H1166.
- Pakkenberg B, Gundersen HJG, Mortensen EL, Lauritzen MJ, Jeune B, Regeur L, West MJ, Schwartz TW. Den normale hjerne: ny viden på flere fronter. Ugeskrift for læger, 1997, 159: 723-727. PMID: 9045457
- Løkkegaard T, Nielsen JE, Hasholt L, Fenger K, Werdelin L, Tranebjaerg L, Lauritzen M, Colding-Jørgensen E, Grønbech-Jensen M, Henriksen OA, Sørensen Machado-Joseph Disease in three Scandinavian Families. J Neurol Sci, 1998, 156: 152-157. doi: 10.1016/s0022-510x(98)00081-1.
- Dreier JP, Körner K, Ebert N, Görner A, Rubin I, Bach T, Lindauer U, Wolf T, Villringer A, Einhäupl K, Lauritzen M and Dirnagl U. Nitric oxide scavenging by hemoglobin or nitric oxide synthase inhibition by N-Nitro-L-Arginine induces cortical spreading ischemia when K+ is increased in the subarachnoid space. J Cereb Blood Flow Metab, 18:978-990, 1998. doi: 10.1097/00004647-199809000-00007.
- Mathiesen C, Caesar K, Akgören N, Lauritzen M. Modification of activity-dependent increases of cerebral blood flow by excitatory synaptic activity and spikes in rat cerebellar cortex. J Physiol, 1998, 512(2) 555-566. doi: 10.1111/j.1469-7793.1998.555be.x.
- Gervil M, Andersen A R, Skriver, E B, Lauritzen M J, Knudsen, F U. Den kliniske værdi af SPECT-scanning hos børn med neurologiske sygdomme. Ugeskrift For Læger, 1998, 48: 6965-6971. PMID: 9846092
- Andersen B, Rössler K, Lauritzen M. Nonspecific Facilitation of Responses to Transcranial Magnetic Stimulation. Muscle and Nerve, 1999, 22: 857-864. doi: 10.1002/(sici)1097-4598(199907)22:7<857::aid-mus7>3.0.co;2-b.
- Zochodne D W, Levy D, Zwiers H, Sun H, Rubin I, Cheng C, Lauritzen M. Evidence for nitric oxide elaboration and nitric oxide synthase activity in the regenerative milieu of proximal stumps from transsected peripheral nerves. Neuroscience, 1999, 91: 1515-1527. doi: 10.1016/s0306-4522(98)00729-5.
- Caesar K, Akgören N, Mathiesen C, Lauritzen M. Modification of activity-dependent increases in cerebellar blood flow by extracellular potassium in anaesthetized rats. J Physiol, 1999, 520. 1: 281-292. doi: 10.1111/j.1469-7793.1999.00281.x.
- Akgören N, Lauritzen M. Functional recruitment of red blood cells to rat brain microcirculation accompanying increased neuronal activity in cerebellar cortex. Neuroreport 1999; 10: 3257-3263. doi: 10.1097/00001756-199911080-00002.
- Lauritzen M. Perfusion-weighted imaging during migraine: spontaneous visual aura and headache [editorial; comment]. Cephalalgia 1999; 19: 697. doi: 10.1046/j.1468-2982.1999.019008697.x.
- Albeck M.J, Taher G, Lauritzen M, Trojaborg W. Diagnostic value of electrophysiological tests in patients with sciatica. Acta Neurol Scand 2000; 101: 249-254. doi: 10.1034/j.1600-0404.2000.101004249.x.
- Mathiesen C, Caesar K, Lauritzen M. Temporal coupling between neuronal activity and blood flow in rat cerebellar cortex as indicated by field potential analysis. J Physiol, 2000; 523: 235-246. doi: 10.1111/j.1469-7793.2000.t01-1-00235.x.
- Thomsen K, Rubin I, Lauritzen M. In vivo mechanisms of acetylcholine-induced vasodilation in rat sciatic nerve. Am J Physiol. 2000; 279: H1044-H1054. doi: 10.1152/ajpheart.2000.279.3.H1044.
- Tomita M, Suzuki N, Hamel E, Busija D, Lauritzen M. Regulation of cerebral microcirculation-update. Keio J Med, 2000; 49: 26-34. doi: 10.2302/kjm.49.26.
- Nielsen AN, Fabricius M, Lauritzen M. Scanning Laser-Doppler Flowmetry of Rat Cerebral Circulation during Cortical Spreading Depression. J Vasc Res, 2000; 37: 513-522. doi: 10.1159/000054084.
- Zochodne DW, Verge VM, Cheng C, Hoke A, Jolley C, Thomsen K, Rubin I, Lauritzen M. Nitric Oxide Synthase Activity and Expression in Experimental Diabetic Neuropathy. J Neuropathol Exp Neurol. 2000; 59: 798-807. doi: 10.1093/jnen/59.9.798.
- Gaspar C, Lopes-Cendes I, Hayes S, Goto J, Arvidsson K, Dias A, Silveira I, Maciel P, Coutinho P, Lima M, Zhou Y X, Soong BW, Watanabe M, Giunti P, Stevanin G, Riess O, Sasaki H, Hsieh M, Nicholson G A, Brunt E, Higgins J J, Lauritzen M, Tranebjaerg L, Volpini V, Wood N, Ranum L, Tsuji S, Brice A, Sequeiros J, and Rouleau G A. Ancestral Origins of the Machado-Joseph Disease Mutation: A Worldwide Haplotype Study. Am J Hum Genet, 2001; 68: 523-529. doi: 10.1086/318184. Epub 2000 Dec 20.
- Nielsen A N and Lauritzen M. Coupling and uncoupling of activity dependent increases of neuronal activity and blood flow in rat somatosensory cortex. J Physiol, 2001; 533.3: 773-785. doi: 10.1111/j.1469-7793.2001.00773.x.
- Lauritzen M. Cortical spreading depression in migraine. Cephalalgia, 2001; 21: 757-760. doi: 10.1111/j.1468-2982.2001.00244.x.
- Lauritzen M. Relationship of spikes, synaptic activity and local changes of cerebral blood flow. J Cereb Blood Flow Metab 2001, 21: 1367-1384. doi: 10.1097/00004647-200112000-00001.
- Broholm H, Brændstrup O and Lauritzen M. Nitric oxide synthase expression of oligodendrogliomas. Clinical Neuropathology, 2001; 20; 6: 233-238. PMID: 11758777
- Gold L, Lauritzen M. Neuronal deactivation explains decreased cerebellar blood flow in response to focal cerebral ischemia or suppressed neocortical function. Proc Nat Acad Sci, (U.S.A), 2002; 99; 11: 7699-7704. doi: 10.1073/pnas.112012499.
- Thomsen K, Rubin I and Lauritzen M. NO-and non-NO-, non-prostanoid-dependent vasodilatation in rat sciatic nerve during maturation and developing experimental diabetic neuropathy. J Physiol, 2002; 543.3; 977-993. doi: 10.1113/jphysiol.2002.023564.
- Thomsen JF, Hansson G-Å, Mikkelsen S and Lauritzen M. Carpal Tunnel syndrome in Repetitive Work: A Follow-Up Study. Am J Indust Med, 2002; 42; 344-353. doi: 10.1002/ajim.10115
- Strong A.J, Fabricius M, Boutelle M.G., Hibbins, S.J., Hopwood S.E., Jones, R, Parkin M.C., Lauritzen M. Spreading and Synchronous Depressions of Cortical Activity in Acutely Injured Human Brain. Stroke, 2002, 2739-2744. doi: 10.1161/01.str.0000043073.69602.09
- Kruse A, Broholm H, Rubin I, Schmidt K., Lauritzen M. Nitric oxide synthase activity in human pituitary adenomas. Acta Neurol Scand 2002; 106: 361-366. doi: 10.1034/j.1600-0404.2002.01138.x.
- Caesar K, Gold L, Lauritzen M. Context sensitivity of activity-dependent increases in cerebral blood flow. Proc Natl Acad Sci (USA) 2003; 100: 4239-4244. doi: 10.1073/pnas.0635075100
- Lauritzen M, Gold L. Brain Function and Neurophysiological Correlates of Signals Used in Functional Neuroimaging. J Neuroscience 2003; 23: 3972-3980. doi: 10.1523/JNEUROSCI.23-10-03972.2003
- Broholm H, Rubin I, Kruse A, Braendstrup O, Schmidt K, Skriver E B & Lauritzen M. Nitric oxide synthase expression and enzymatic activity in human brain tumors. Clin Neuropathol 2003; 22: 273-281. PMID: 14672505
- Caesar K, Thomsen K & Lauritzen M. Dissociation of spikes, synaptic activity, and activity-dependent increments in rat cerebellar blood flow by tonic synaptic inhibition. Proc Natl Acad Sci (USA) 2003; 100: 16000-16005. doi: 10.1073/pnas.2635195100
- Broholm H, Andersen B, Wanscher B, Frederiksen J L, Rubin I, Pakkenberg B, Larsson H B W, Lauritzen M. Nitric oxide synthase expression and enzymatic activity in multiple sclerosis. Acta Neurol Scand 2004; 109: 2610-270. doi: 10.1111/j.1600-0404.2004.00207.x.
- Piilgaard H, Lauritzen M. Contribution of somatosensory cortex to evoked cerebellar blood flow responses. Neuroreport 2004; 15: 695-698. doi: 10.1097/00001756-200403220-00024
- Enager P, Gold L, Lauritzen M. Impaired neurovascular Coupling by Transhemispheric Diaschisis in Rat Cerebral Cortex. J Cereb Blood Flow Metab 2004, 24: 713-719. doi: 10.1097/01.WCB.0000121233.63924.41.
- Thomsen K, Offenhauser N, Lauritzen M. Principle neuron spiking: neither necessary nor sufficient for cerebral blood flow at rest or during activation in rat cerebellum. J Physiol, London, 2004, 360:181-189. doi: 10.1113/jphysiol.2004.068072.
- Lauritzen M. Reading vascular changes in brain imaging: Is dendritic calcium the key. Nature Rev Neurosci, 2005, 6(1) 77-85. doi: 10.1038/nrn1589.
- Parkin M, Hopwood S, Jones D, Hashemi P, Landolt H, Fabricius M, Lauritzen M, Boutelle M, Strong A. Dynamic changes in brain glucose and lactate in pericontusional areas of the human cerebral cortex, monitored with rapid sampling on-line microdialysis: relationship with depolarisation-like events. J Cereb Blood Flow Metab 2005, 25:402-413. doi: 10.1038/sj.jcbfm.9600051.
- Roed HG, Langkilde A, Sellebjerg F, Lauritzen M, Bang P, Mørup A, Frederiksen J. A double-blind, randomized, placebo-controlled trial of intravenous immunoglobulin treatment in acute optic neuritis. Neurology 2005, 64(5):804-810. DOI: 1212/01.WNL.0000152873.82631.B3
- Roed H, Frederiksen J, Langkilde A, Sørensen TL, Lauritzen M, Sellebjerg F. Systemic Tcell activation in acute clinically isolated optic neuritis. J Neuroimmunol. 2005, May;162(1-2):165-72. doi: 10.1016/j.jneuroim.2005.02.002
- Offenhauser N, Thomsen K, Caesar K, Lauritzen M. Activity-induced tissue oxygenation changes in rat cerebellar cortex: Interplay of postsynaptic activation and blood flow. J Physiol, 2005, 565(1):279-294. doi: 10.1113/jphysiol.2005.082776.
- Fabricius M, Fuhr S, Bhatia R, Boutelle M, Hashemi P, Strong AJ, Lauritzen M. Cortical spreading depression and peri-infarct depolarization in acutely injured human cerebral cortex. Brain 2006, 129(Pt.3):778-90. doi: 10.1093/brain/awh716.
- Hoffmeyer HW, Enager P, Thomsen KJ, Lauritzen MJ. Nonlinear neurovascular coupling in rat sensory cortex by activation of transcallosal fibers. J Cereb Blood Flow Metab. 2007, 27: 575-587. doi: 10.1038/sj.jcbfm.9600372
- Dreier JP, Woitzik J, Fabricius M, Bhatia R, Major S, Drenckhahn C, Lehmann TN, Sarrafzadeh A, Willumsen L, Hartings JA, Sakowitz OW, Seemann JH, Thieme A, Lauritzen M. Strong AJ. Delayed ischaemic neurological deficits after subarachnoid haemorrhage are associated with clusters of spreading depolarizations. Brain. 2006, 129: 3224-3237. doi: 10.1093/brain/awl297
- Caesar K, Offenhauser N, Lauritzen M. Gamma-aminobutyric acid modulates local brain oxygen consumption and blood flow in rat cerebellar cortex. Journal of Cerebral Blood Flow & Metabolism. 2008, 28: 906-915. doi: 10.1038/sj.jcbfm.9600581.
- Caesar K, Hashemi P, Douhou A, Bonvento G, Boutelle MG, Walls AB, Lauritzen M. Glutamate receptor-dependent increments in lactate, glucose and oxygen metabolism evoked in rat cerebellum in vivo. J. Physiol. 2008, 586.5, 1337-1349. doi: 10.1113/jphysiol.2007.144154.
- Sander M, Avlund K, Lauritzen M, Gottlieb T, Halliwell B, Stevnsner T, Wewer U, Bohr VA.
Aging-from molecules to populations. Mech Ageing Dev. 2008 Oct;129(10):614-23. doi: 10.1016/j.mad.2008.08.002.
- Lauritzen M. On the neural basis of fMRI signals. Clin Neurophysiol. 2008 Apr;119(4):729-30. doi: 0.1016/j.clinph.2007.10.025. Epub 2007 Dec 27.
- Fabricius M, Fuhr S, Willumsen L, Dreier JP, Bhatia R, Boutelle MG, Hartings JA, Bullock R, Strong AJ, Lauritzen M. Association of seizures with cortical spreading depression and peri-infarct depolarisations in the acutely injured human brain. Clinical Neurophysiology. 2008, 119: 1973-1984. doi: 10.1016/j.clinph.2008.05.025
- Kruuse C, Hansen AE, Larsson HB, Lauritzen M, Rostrup E. Cerebral haemodynamic response or excitability is not affected by sildenafil. J Cereb Blood Flow Metab. 2009 Apr; 29(4):830-9. doi: 10.1038/jcbfm.2009.10.
- Enager P, Piilgaard H, Offenhauser N, Kocharyan A, Fernandes P, Hamel E, Lauritzen M. Pathway-specific variations in neurovascular and neurometabolic coupling in rat primary somatosensory cortex. J Cereb Blood Flow Metab. 2009 May;29(5):976-86. doi: 10.1038/jcbfm.2009.23.
- Rasmussen, T, Rathlau NH, Lauritzen M. Modeling neuro-vascular coupling in rat cerebellum: characterization of deviations from linearity. 2009 Mar 1;45(1):96-108. doi: 10.1016/j.neuroimage.2008.10.023.
- Dreier JP, Major S, Manning A, Woitzik J, Drenckhahn C, Steinbrink J, Tolias C, Oliveira-Ferreira AI, Fabricius M, Hartings JA, Vajkoczy P, Lauritzen M, Dirnagl U, Bohner G, Strong AJ; COSBID study group. Cortical spreading ischaemia is a novel process involved in ischaemic damage in patients with aneurysmal subarachnoid haemorrhage. 2009 Jul;132(Pt 7):1866-81. doi: 10.1093/brain/awp102.
- Piilgaard H, Lauritzen M. Persistent increase in oxygen consumption and impaired neurovascular coupling after spreading depression in rat neocortex. J Cereb Blood Flow Metab. 2009 Sep;29(9):1517-27. doi: 10.1038/jcbfm.2009.73
- Thomsen K, Piilgaard H, Gjedde A, Bonvento G, Lauritzen M. Principal cell spiking, postsynaptic excitation, and oxygen consumption in the rat cerebellar cortex. J Neurophysiol. 2009 Sep;102(3):1503-12. doi: 10.1152/jn.00289.2009.
- Eriksen N, Rostrup E, Andersen K, Lauritzen MJ, Fabricius M, Larsen VA, Dreier JP, Strong AJ, Hartings JA, Pakkenberg B. Application of stereological estimates in patients with severe head injuries using CT and MR scanning images. Br J Radiol. 2010; 83:307-317. doi: 10.1259/bjr/18575224.
- Attwell D, Buchan AM, Charpak S, Lauritzen M, MacVicar BA, Newman EA. Glial and neuronal control of brain blood flow. Nature. 2010, 468:232-243. doi: 10.1038/nature09613.
- Nieto-Gonzalez JL, Moser J, Lauritzen M, Schmitt-John T, Jensen K. Reduced GABAergic Inhibition Explains Cortical Hyperexcitability in the Wobbler Mouse Model of ALS. Cereb Cortex. 2011, 21:625-635. doi: 10.1093/cercor/bhq134.
- Lauritzen M, Dreier JP, Fabricius, M, Hartings, J, Graf R, Strong AJ. Clinical relevance of cortical spreading depression in neurological disorders: migraine, malignant stroke, subarachnoid & intracranial hemorrhage, and traumatic brain injury. J Cereb Blood Flow Metab, 2011, 31:17-35. doi: 10.1038/jcbfm.2010.191.
- Piilgaard H, Lauritzen M. Cyclosporine A, FK506, and NIM811 ameliorate prolonged CBF reduction and impaired neurovascular coupling after cortical spreading depression. J Cereb Blood Flow Metab, 2011, 31: 1588-1598. doi: 10.1038/jcbfm.2011.28.
- Mathiesen, C, Caesar, K, Thomsen, K, Hoogland, TM, Witgen, BM, Brazhe, A and Lauritzen M. Activity-dependent Increases in Local Oxygen Consumption Correlate with Postsynaptic Currents in the Mouse Cerebellum In Vivo. Journal of Neuroscience. 14; 31(50):18327-37, 20. doi: 10.1523/JNEUROSCI.4526-11.2011.
- Schaefer, K, Blankenburg, F, Kupers, R, Grüner, JM, Law, I, Lauritzen, M and Larsson HB. Negative BOLD signal changes in ipsilateral primary somatosensory cortex are associated with perfusion decreases and behavioral evidence for functional inhibition. 2012 Feb 15;59(4):3119-27. doi: 10.1016/j.neuroimage.2011.11.085.
- Lauritzen, M, Mathiesen, C, Schäfer, K and Thomsen, KJ. Neuronal inhibition and excitation, and the dichotomic control of brain hemodynamic and oxygen responses. Neuroimage. 2012, Aug 15;62(2):1040-50. doi: 10.1016/j.neuroimage.2012.01.040.
- Pedersen GL, Rasmussen SB, Gyllenborg J, Benedek K, Lauritzen M. Prognostic value of periodic electroencephalographic discharges for neurological patients with profound disturbances of consciousness. Clin Neurophysiol. 2013 Jan;124(1):44-51. doi: 10.1016/j.clinph.2012.06.010.
- Fordsmann J, Ko R, Choi H, Thomsen K, Witgen B, Mathiesen C, Lønstrup M, Henning Piilgaard, MacVicar B, Lauritzen M. Increased 20-HETE synthesis explains reduced cerebral blood flow but not impaired neurovascular coupling after cortical spreading depression in rat cerebral cortex. J Neuroscience, 2013;33:2562-2570. doi: 10.1523/JNEUROSCI.2308-12.2013.
- Lind BL, Brazhe AR, Jessen SB, Tan FC, Lauritzen MJ: Rapid stimulus-evoked astrocyte Ca2+ elevations and hemodynamic responses in mouse somatosensory cortex in vivo. Proc Natl Acad Sci U S A 2013;110:E4678-E4687. doi: 10.1073/pnas.1310065110.
- Mathiesen C, Brazhe A, Thomsen K, Lauritzen M: Spontaneous calcium waves in Bergman glia increase with age and hypoxia and may reduce tissue oxygen. J Cereb Blood Flow Metab 2013; 33:161-169. doi: 10.1038/jcbfm.2012.175.
- Brazhe A, Mathiesen C, Lauritzen M: Multiscale vision model highlights spontaneous glial calcium waves recorded by 2-photon imaging in brain tissue. Neuroimage 2013;68:192-202. doi: 10.1016/j.neuroimage.2012.11.024.
- Brazhe A, Mathiesen C, Lind B, Rubin A, Lauritzen M: Multiscale vision model for event detection and reconstruction in two-photon imaging data. Neurophotonics 2014;1:011012. doi: 10.1117/1.NPh.1.1.011012.
- Lindqvist JK, Lauritzen M, Laursen H, Benedek K: [Myopathy causing acute tetraparesis in the intensive care unit.]. Ugeskr Laeger 2014;176.
- Hansen NL, Lauritzen M, Mortensen EL, Osler M, Avlund K, Fagerlund B, Rostrup E: Subclinical cognitive decline in middle-age is associated with reduced task-induced deactivation of the brain's default mode network. Hum Brain Map 2014;35:4488-4498. doi: 10.1002/hbm.22489.
- Sorensen CE, Larsen JO, Reibel J, Lauritzen M, Mortensen EL, Osler M, Pedersen AM: Associations between xerostomia, histopathological alterations, and autonomic innervation of labial salivary glands in men in late midlife. Exp Gerontol 2014;57:211-217. doi: 10.1016/j.exger.2014.06.004.
- Hall CN, Reynell C, Gesslein B, Hamilton NB, Mishra A, Sutherland BA, O'Farrell FM, Buchan AM, Lauritzen M**, Attwell D**: Capillary pericytes regulate cerebral blood flow in health and disease. Nature 2014;508:55-60. doi: 10.1038/nature13165. **Co-corresponding authors*
- Jessen SB, Brazhe A, Lind BL, Mathiesen C, Thomsen K, Jensen K, Lauritzen M: GABAA Receptor-Mediated Bidirectional Control of Synaptic Activity, Intracellular Ca2+, Cerebral Blood Flow, and Oxygen Consumption in Mouse Somatosensory Cortex In Vivo. Cereb Cortex 2015;25:2594-2609. doi: 10.1093/cercor/bhu058.
- Ayata C, Lauritzen M: Spreading Depression, Spreading Depolarizations, and the Cerebral Vasculature. Physiol Rev 2015;95:953-993. doi: 10.1152/physrev.00027.2014.
- Desler C, Frederiksen JH, Angleys M, Maynard S, Keijzers G, Fagerlund B, Mortensen EL, Osler M, Lauritzen M, Bohr VA, Rasmussen LJ. Increased deoxythymidine triphosphate levels is a feature of relative cognitive decline. Mitochondrion. 2015 Sep 25;25:34-37. doi: 10.1016/j.mito.2015.09.002.
- Waller KL, Mortensen EL, Avlund K, Osler M, Fagerlund B, Lauritzen M, Jennum P. Subjective sleep quality and daytime sleepiness in late midlife and their association with age-related changes in cognition. Sleep Med. 2016, 165-173. doi: 10.1016/j.sleep.2015.01.004.
- Jessen SB, Mathiesen C, Lind BL, Lauritzen M. Interneuron deficit associates attenuated network synchronization to mismatch of energy supply and demand in aging mouse brains. Cereb Cortex 2017, 27(1): 646-659. doi: 10.1093/cercor/bhv261.
- Kucharz K, Søndergaard Rasmussen I, Bach A, Strømgaard K, Lauritzen M. PSD-95 uncoupling from NMDA receptors by Tat-N-dimer ameliorates neuronal depolarisation in cortical spreading depression. J Cereb Blood Flow Metab. 2017, 37(5) 1820-1828. doi: 10.1177/0271678X16645595.
- Sørensen CE, Tritsaris K, Reibel J, Lauritzen M, Mortensen EL, Osler M, Pedersen AM. Elevated p16ink4a Expression in Human Labial Salivary Glands as a Potential Correlate of Cognitive Aging in Late Midlife. PLoS One. 2016 Mar 30;11(3):e0152612. doi: 10.1371/journal.pone.0152612.
- Benedek K, Berényi A, Gombkötő P, Piilgaard H, Lauritzen M. Neocortical gamma oscillations in idiopathic generalized epilepsy. Epilepsia. 2016 May;57(5):796-804. doi: 10.1111/epi.13355. Epub 2016 Mar 21. doi: 10.1111/epi.13355.
- Bøttger P, Glerup S, Gesslein B, Illarionova NB, Isaksen TJ, Heuck A, Clausen BH, Füchtbauer EM, Gramsbergen JB, Gunnarson E, Aperia A, Lauritzen M, Lambertsen KL, Nissen P, Lykke-Hartmann K. Glutamate-system defects behind psychiatric manifestations in a familial hemiplegic migraine type 2 disease-mutation mouse model. Sci Rep. 2016, 25;6:22047. doi: 10.1038/srep22047.
- Waller KL, Mortensen EL, Avlund K, Fagerlund B, Lauritzen M, Gammeltoft S, Jennum P. Melatonin and cortisol profiles in late midlife and their association with age-related changes in cognition. Nat Sci Sleep. 2016 Jan 21;8:47-53. oi: 10.2147/NSS.S75946.
- Sørensen L, Igel C, Liv Hansen N, Osler M, Lauritzen M, Rostrup E, Nielsen M; Alzheimer's Disease Neuroimaging Initiative and the Australian Imaging Biomarkers and Lifestyle Flagship Study of Ageing. Early detection of Alzheimer's disease using MRI hippocampal texture. Hum Brain Mapp. 2016 Mar;37(3):1148-61. doi: 10.1002/hbm.23091.
- Waller KL, Mortensen EL, Avlund K, Osler M, Fagerlund B, Lauritzen M, Jennum P. Subjective sleep quality and daytime sleepiness in late midlife and their association with age-related changes in cognition. Sleep Med. 2016 Jan;17:165-73. doi: 10.1016/j.sleep.2015.01.004.
- Khennouf L, Gesslein B, Lind BL, van den Maagdenberg AM, Lauritzen M. Activity-dependent calcium, oxygen and vascular responses in a mouse model of familial hemiplegic migraine type 1. Ann Neurol. 2016 Aug;80(2):219-32. doi: 10.1002/ana.24707.
- Lauritzen M, Strong AJ. 'Spreading depression of Leão' and its emerging relevance to acute brain injury in humans. J Cereb Blood Flow Metab. 2017, 37(5): 1553-1570. doi: 10.1177/0271678X16657092.
- Hartings JA, Shuttleworth CW, Kirov SA, Ayata C, Hinzman JM, Foreman B, Andrew RD, Boutelle MG, Brennan KC, Carlson AP, Dahlem MA, Drenckhahn C, Dohmen C, Fabricius M, Farkas E, Feuerstein D, Graf R, Helbok R, Lauritzen M, Major S, Oliveira-Ferreira AI, Richter F, Rosenthal ES, Sakowitz OW, Sánchez-Porras R, Santos E, Schöll M, Strong AJ, Urbach A, Westover MB, Winkler MK, Witte OW, Woitzik J, Dreier JP. The continuum of spreading depolarizations in acute cortical lesion development: Examining Leão's legacy. J Cereb Blood Flow Metab. 37(5): 1571-1594. doi: 10.1177/0271678X16654495.
- Dreier JP, Fabricius M, Ayata C, Sakowitz OW, William Shuttleworth C, Dohmen C, Graf R, Vajkoczy P, Helbok R, Suzuki M, Schiefecker AJ, Major S, Winkler MK, Kang EJ, Milakara D, Oliveira-Ferreira AI, Reiffurth C, Revankar GS, Sugimoto K, Dengler NF, Hecht N, Foreman B, Feyen B, Kondziella D, Friberg CK, Piilgaard H, Rosenthal ES, Westover MB, Maslarova A, Santos E, Hertle D, Sánchez-Porras R, Jewell SL, Balança B, Platz J, Hinzman JM, Lückl J, Schoknecht K, Schöll M, Drenckhahn C, Feuerstein D, Eriksen N, Horst V, Bretz JS, Jahnke P, Scheel M, Bohner G, Rostrup E, Pakkenberg B, Heinemann U, Claassen J, Carlson AP, Kowoll CM, Lublinsky S, Chassidim Y, Shelef I, Friedman A, Brinker G, Reiner M, Kirov SA, Andrew RD, Farkas E, Güresir E, Vatter H, Chung LS, Brennan KC, Lieutaud T, Marinesco S, Maas AI, Sahuquillo J, Dahlem MA, Richter F, Herreras O, Boutelle MG, Okonkwo DO, Bullock MR, Witte OW, Martus P, van den Maagdenberg AM, Ferrari MD, Dijkhuizen RM, Shutter LA, Andaluz N, Schulte AP, MacVicar B, Watanabe T, Woitzik J, Lauritzen M, Strong AJ, Hartings JA. Recording, analysis, and interpretation of spreading depolarizations in neurointensive care: Review and recommendations of the COSBID research group. J Cereb Blood Flow Metab. 2017, 37(5): 1595-1625. doi: 10.1177/0271678X16654496.
- Sutherland BA, Fordsmann JC, Martin C, Neuhaus AA, Witgen BM, Piilgaard H, Lønstrup M, Couch Y, Sibson NR, Lauritzen M, Buchan AM. Multi-modal assessment of neurovascular coupling during cerebral ischaemia and reperfusion using remote middle cerebral artery occlusion. J Cereb Blood Flow Metab. 2017, 37(7) 2494-2508. doi: 10.1177/0271678X16669512.
- Howarth C, Sutherland B, Choi HB, Martin C, Lind BL, Khennouf L, LeDue JM, Janelle MP, Ko RWY, Ellis-Davies G, Lauritzen M, Sibson NR, Buchan AM, Macvicar BA. A critical role for astrocytes in hypercapnic vasodilation in brain. J Neuroscience 37 (9), 2017, 2403-2414. doi: 10.1523/JNEUROSCI.0005-16.2016.
- Horwitz A, Thomsen MD, Wiegand I, Horwitz H, Klemp M, Nikolic M, Rask L, Lauritzen M, Benedek K. Visual steady state in relation to age and cognitive function. PloS one 12 (2), 2017, e0171859. doi: 10.1371/journal.pone.0171859.
- Horwitz A, Mortensen EL, Osler M, Fagerlund B, Lauritzen M, Benedek. Passive Double-Sensory Evoked Coherence Correlates with Long-Term Memory Capacity. Front Hum Neurosci. 2017 Dec 14;11:598. doi: 10.3389/fnhum.2017.00598.
- Rask L, Bendix L, Harbo M, Fagerlund B, Mortensen EL, Lauritzen MJ, Osler M. Cognitive Change during the Life Course and Leukocyte Telomere Length in Late Middle-Aged Men. Front Aging Neurosci. 2016 Dec 9;8:300. doi: 10.3389/fnagi.2016.00300. doi: 10.3389/fnagi.2016.00300.
- OM Henriksen, NL Hansen, M Osler, EL Mortensen, DM Hallam, Pedersen ET, Chappell M, Lauritzen MJ, Rostrup E. Sub-Clinical Cognitive Decline and Resting Cerebral Blood Flow in Middle Aged Men PloS one 12 (1), 2017, e0169912. doi: 10.1371/journal.pone.0169912.
- Thomsen K, Yokota T, Hasan-Olive MM, Sherazi N, Fakouri NM, Desler C, Regnell CE, Larsen S, Rasmussen LJ, Dela F, Bergersen LH, Lauritzen M. Initial brain aging: heterogeneity of mitochondrial size is associated with decline in complex I-linked respiration in cortex and hippocampus. Neurobiol Aging. 2017 Aug 12. pii: S0197-4580(17)30259-2. doi: 10.1016/j.neurobiolaging.2017.08.004.
- Lind BL, Jessen SB, Lønstrup M, Charlène J, Bonvento G, Lauritzen M. Fast Ca2+ responses in astrocyte end-feet and neurovascular coupling in mice. 2018 Feb;66(2):348-358. doi: 10.1002/glia.23246. Epub 2017 Oct 23. doi: 10.1002/glia.23246.
- Fakouri M, Durhuus J, Regnell C, Angleys C, Desler C, Hasan M, Martín-Pardillos A, Tsaalbi-Shtylik A, Thomsen K, Lauritzen M, Wind N, Bergersen LH, Rasmussen L. Rev1 contributes to proper mitochondrial function via the PARP-NAD+-SIRT1-PGC1α axis. Sci Rep. 2017 Oct 2;7(1):12480. doi: 10.1038/s41598-017-12662-3. doi: 10.1038/s41598-017-12662-3.
- Cnossen MC, Huijben JA, van der Jagt M, Volovici V, van Essen T, Polinder S, Nelson D, Ercole A, Stocchetti N, Citerio G, Peul WC, Maas AIR, Menon D, Steyerberg EW, Lingsma HF; CENTER-TBI investigators. Variation in monitoring and treatment policies for intracranial hypertension in traumatic brain injury: a survey in 66 neurotrauma centers participating in the CENTER-TBI study. Crit Care. 2017 Sep 6;21(1):233. doi: 10.1186/s13054-017-1816-9.
- Huijben JA, van der Jagt M, Cnossen MC, Kruip MJHA, Haitsma IK, Stocchetti N, Maas AIR, Menon DK, Ercole A, Maegele M, Stanworth SJ, Citerio G, Polinder S, Steyerberg EW, Lingsma HF; CENTER-TBI Investigators and Participants. Variation in Blood Transfusion and Coagulation Management in Traumatic Brain Injury at the Intensive Care Unit: A Survey in 66 Neurotrauma Centers Participating in the Collaborative European NeuroTrauma Effectiveness Research in Traumatic Brain Injury Study. .J Neurotrauma. 2018 Jan 15;35(2):323-332. doi: 10.1089/neu.2017.5194.
- Wiegand I, Lauritzen MJ, Osler M, Mortensen EL, Rostrup E, Rask L, Richard N, Horwitz A, Benedek K, Vangkilde S, Petersen A. EEG correlates of visual short-term memory in older age vary with adult lifespan cognitive development. Neurobiol Aging. 2017 Oct 31;62:210-220. doi: 10.1016/j.neurobiolaging.2017.10.018.
- Sorensen CE, Hansen NL, Mortensen EL, Lauritzen M, Osler M, Pedersen AML. Hyposalivation and Poor Dental Health Status Are Potential Correlates of Age-Related Cognitive Decline in Late Midlife in Danish Men. Aging Neurosci., 2018. doi.org/10.3389/fnagi.2018.00010
- Christensen RK, Delgado-Lezama R, Russo RE, Lind BL, Alcocer EL, Rath MF, Fabbiani G, Schmitt N, Lauritzen M, Petersen AV, Carlsen EM, Perrier JF Spinal dorsal horn astrocytes release GABA in response to synaptic activation. J Physiol. 2018 Oct;596(20):4983-4994. doi: 10.1113/JP276562.
- Kucharz K, Lauritzen M. NMDA receptor-dependent endoplasmic reticulum fission-fusion accompanies rises in synaptic activity and cortical spreading depolarization waves. Brain. 2018 Apr 1;141(4):1049-1062. doi: 10.1093/brain/awy036.
- Brazhe NA, Thomsen K, Lønstrup M, Brazhe AR, Nikelshparg EI, Maksimov GV, Lauritzen M, Sosnovtseva O. Monitoring of blood oxygenation in brain by resonance Raman spectroscopy. J Biophotonics. 2018 Mar 30:e201700311. doi: 10.1002/jbio.201700311.
- Khennouf L, Gesslein B, Brazhe A, Octeau JC, Kutuzov N, Khakh BS, Lauritzen M. Active role of capillary pericytes during stimulation-induced activity and spreading depolarization. Brain 2018, July, 141 (7); 2032–2046. https://doi.org/10.1093/brain/awy143
- Cai C, Fordsmann J, Jensen SH, Hald BO, Gesslein B, Lønstrup M, Brodin B, Lauritzen M. Conducted vascular responses in brain capillaries by synaptic activity and ATP in mouse cerebral cortex. Proc Natl Acad Sci U S A. 2018 Jun 19;115(25):E5796-E5804. doi:.1073/pnas.1707702115.
- Fordsmann JC, Murmu RP, Cai C, Brazhe A, Thomsen KJ, Zambach SA, Lønstrup M, Lind BL, Lauritzen Spontaneous astrocytic Ca2+ activity abounds in electrically suppressed ischemic penumbra of aged mice. Glia. 2019 Jan;67(1):37-52. doi: 10.1002/glia.23506.
- Kutuzov N, Flyvbjerg H, Lauritzen M. Contributions of the glycocalyx, endothelium, and extravascular compartment to the blood-brain barrier. Proc Natl Acad Sci U S A. 2018 Oct 2;115(40):E9429-E9438. doi: 10.1073/pnas.1802155115
- Eriksen N, Rostrup E, Fabricius M, Scheel M, Major S, Winkler MKL, Bohner G, Santos E, Sakowitz OW, Kola V, Reiffurth C, Hartings JA, Vajkoczy P, Woitzik J, Martus P, Lauritzen M, Pakkenberg B, Dreier JP. Early focal brain injury after subarachnoid hemorrhage correlates with spreading depolarizations. 2019 Jan 22;92(4):e326-e341. doi: 10.1212/WNL.0000000000006814.
- Eriksen N, Pakkenberg B, Rostrup E, Okonkwo DO, Mathern B, Shutter LA, Strong AJ, Woitzik J, Pahl C, Dreier JP, Martus P, Lauritzen MJ, Fabricius M, Hartings JA. Neurostereologic Lesion Volumes and Spreading Depolarizations in Severe Traumatic Brain Injury Patients: A Pilot Study. Neurocrit Care. 2019 Jun;30(3):557-568. doi: 10.1007/s12028-019-00692-w.
- Waser M, Lauritzen MJ, Fagerlund B, Osler M, Mortensen EL, Sørensen HBD, Jennum P. Sleep efficiency and neurophysiological patterns in middle-aged men are associated with cognitive change over their adult life course. J Sleep Res. 2018 Nov 12:e12793. doi: 10.1111/jsr.12793.
- Nielsen RM, Urdanibia-Centelles O, Vedel-Larsen E, Thomsen KJ, Møller K, Olsen KS, Lauritsen AØ, Eddelien HS, Lauritzen M, Benedek K. Continuous EEG Monitoring in a Consecutive Patient Cohort with Sepsis and Delirium. Neurocrit Care. 2019 Mar 19. doi: 10.1007/s12028-019-00703-w.
- Jensen AJ, Lauritzen M. Diagnosing sports-related concussion in football players. Ugeskr. Laeger. 2019 Jun 17;181(25). pii: V12180883, Danish. PMID: 31267946
- Cai C, Zambach SA, Fordsmann JC, Lønstrup M, Thomsen KJ, Jensen AGK, Lauritzen M. In Vivo Three-Dimensional Two-Photon Microscopy to Study Conducted Vascular Responses by Local ATP Ejection Using a Glass Micro-Pipette. J Vis Exp. 2019 Jun 7;(148). doi: 10.3791/59286.
- Horwitz A, Klemp M, Horwitz H, Thomsen MD, Rostrup E, Mortensen EL, Osler M, Lauritzen M, Benedek K. Brain Responses to Passive Sensory Stimulation Correlate With Intelligence. Front Aging Neurosci. 2019 Aug 14;11:201. doi: 10.3389/fnagi.2019.00201.
- Zarnani K, Nichols TE, Alfaro-Almagro F, Fagerlund B, Lauritzen M, Rostrup E, Smith SM. Discovering markers of healthy aging: a prospective study in a Danish male birth cohort. Aging (Albany NY). 2019 Aug 26;11(16):5943-5974. doi: 10.18632/aging.102151.
- Dahlqvist, M., Thomsen K, Postnov D, Lauritzen, M. Modification of oxygen consumption and blood flow in mouse somatosensory cortex by cell-type-specific neuronal activity. Journal of Cerebral Blood Flow and Metabolism, 2019, 2019 Oct 23:271678X19882787. doi: 10.1177/0271678X19882787
- Murmu RP, Fordsmann JC, Cai C, Brazhe A, Thomsen KJ, Lauritzen M. Sensory Stimulation-Induced Astrocytic Calcium Signaling in Electrically Silent Ischemic Penumbra. Front Aging Neurosci. 2019 Aug 21;11:223. doi: 10.3389/fnagi.2019.00223.
- Grubb S, Lauritzen M. Brain waves drive fluid flow during deep sleep. Invited perspective paper. Science. 2019 Nov 1;366(6465):572-573. doi: 10.1126/science.aaz5191.
- Janiurek MM, Soylu-Kucharz R, Christoffersen C, Kucharz K, Lauritzen M. Apolipoprotein M-bound sphingosine-1-phosphate regulates blood–brain barrier paracellular permeability and transcytosis. 2019 Nov 25;8. pii: e49405. doi: 10.7554/eLife.49405.
- Grubb S, Cai C, Hald BO, Khennouf L, Fordsmann J, Murmu R, Jensen AKG, Zambach S, Lauritzen M. Precapillary sphincters maintain perfusion in the cerebral cortex. Nat Commun. 2020 Jan 20;11(1):395. doi: 10.1038/s41467-020-14330-z.
- Richard N, Nikolic M, Mortensen EL, Osler M, Lauritzen M, Benedek K. Steady-state visual evoked potential temporal dynamics reveal correlatesof cognitive decline. Clin Neurophysiol 2020: 10.1016/j.clinph.2020.01.010. doi: 10.1016/j.clinph.2020.01.010.
- Kristensen M, Kucharz K, Fernandes E, Strømgaard K, Schallburg Nielsen M, Cederberg Helms HC, Bach A, Ulrikkaholm Tofte-Hansen M, Garcia B, Lauritzen M, Brodin B. Conjugation of Therapeutic PSD-95 Inhibitors to the Cell-Penetrating Peptide Tat Affects Blood-Brain Barrier Adherence, Uptake, and Permeation. Pharmaceutics. 2020 Jul 14;12(7): E661. doi: 10.3390/pharmaceutics12070661.
- Zarnani K, Smith SM, Alfaro-Almagro F, Fagerlund B, Lauritzen M, Rostrup E, Nichols TE. Discovering correlates of age-related decline in a healthy late-midlife male birth cohort. Aging (Albany NY) 2020 Sep 15; 12(17): 16709–16743. doi: 10.18632/aging.
- Grubb S, Lauritzen M, Aalkjær C. Brain capillary pericytes and neurovascular coupling. Comp Biochem Physiol A Mol Integr Physiol. 2021 Jan 6;254:110893. doi: 10.1016/j.cbpa.2020.110893.
- Urdanibia-Centelles O, Nielsen RM, Rostrup E, Vedel-Larsen E, Thomsen K, Nikolic M, Johnsen B, Møller K, Lauritzen M, Benedek K. Automatic cEEG signal analysis for diagnosis of delirium in patients with sepsis. Clin Neurophysiol. 2021 Sep;132(9):2075-2082. doi: 10.1016/j.clinph.2021.05.013. Epub 2021 Jun 9.PMID: 34284242
- Zambach SA, Changsi C, Helms HC, Hald BO, Dongd Y, Fordsmann JC, Nielsen RN, Hud J, Lønstrup M, Brodin B, Lauritzen M. Precapillary sphincters and pericytes at first-order capillaries as key regulators for brain capillary perfusion. Proc Natl Acad Sci U S A 2021, doi: 10.1073/pnas.2023749118.
- Hørlyck S, Cai C, Helms HCC, Lauritzen M, Brodin B. ATP induces contraction of cultured brain capillary pericytes via activation of P2Y-type purinergic receptors. Am J Physiol Heart Circ Physiol. 2021 Feb 1;320(2):H699-H712. doi: 10.1152/ajpheart.00560.2020.
- Kucharz K, Kristensen K, Johnsen KB, Lund MA, Lønstrup M, Moos T, Andresen TL, Lauritzen M. Post-capillary venules are the key locus for transcytosis-mediated brain delivery of therapeutic Nat Comm 2021 Jul 5;12(1):4121. doi: 10.1038/s41467-021-24323-1.
- Mächler P, Mateo C, Thunemann M, Fomin-Thunemann N, Doran PR, Kilic K, Sencan I, Desjardins M, Uhlirova H, Yaseen MA, Boas DA, Linninger AA, Vergassola M, Yu X, Lewis L, Polimeni J, Rosen BR, Sakadžić S, Buxton RB, Lauritzen M, Kleinfeld D, Devor A. New Avenues for Physiological Underpinning of Functional Imaging Signals with Novel Microscopic Imaging Tools. Curr Opin Biomed Eng. 2021 Jun;18:100273. doi: 10.1016/j.cobme.2021.100273.
- Vestergaard MB, Lindberg U, Knudsen MH, Urdanibia-Centelles O, Bakhtiari A, Mortensen EL, Osler M, Fagerlund B, Benedek K, Lauritzen M, Larsson HBW. Subclinical cognitive deficits are associated with reduced cerebrovascular response to visual stimulation in mid-sixties men. Geroscience. 2022 Jun 1. doi: 10.1007/s11357-022-00596-2.
- Kucharz K, Kutuzov N, Zhukov O, Mathiesen Janiurek M, Lauritzen M. Pharm Res. Shedding Light on the Blood-Brain Barrier Transport with Two-Photon Microscopy In Vivo. 2022 May 16. doi: 10.1007/s11095-022-03266-2.
- Wiggers A, Ashina H, Hadjikhani N, Sagare A, Zlokovic B, Lauritzen M, Ashina M. Brain barriers and their potential role in migraine pathophysiology. The Journal of Headache and Pain 2022. 23 (1), 1-10. doi: 10.1186/s10194-021-01365-w.
- Thomsen MS, Johnsen KB, Kucharz K, Lauritzen M, Moos T. Blood-Brain Barrier Transport of Transferrin Receptor-Targeted Nanoparticles. Pharmaceutics 2022. 14:2237-2247. doi: 10.3390/pharmaceutics14102237.
- Bakhtiari A, Vestergaard MB, Benedek K, Fagerlund B, Mortensen EL, Osler M, Lauritzen M, Larsson HBW, Lindberg U. Changes in hippocampal volume during a preceding ten-year period do not correlate with cognitive performance and hippocampal blood‒brain barrier permeability in cognitively normal late-middle-aged men. Geroscience 2023 Apr;45(2):1161-1175. doi: 10.1007/s11357-022-00712-2.
- Cai C, Zambach S, Grubb S, Tao L, He C, Lind BL, Thomsen KJ, Zhang X, Hald BO, Nielsen RM, Kim K, Devor A, Lønstrup M, Lauritzen MJ. Impaired dynamics of precapillary sphincters and pericytes at first-order capillaries predict reduced neurovascular function in the aging mouse brain. Nat Aging. 2023 Feb;3(2):173-184. doi: 10.1038/s43587-022-00354-1.
- Liu X, Whalen AJ, Ryu SB, Lee SW, Fried SI, Kim K, Cai C, Lauritzen M, Bertram N, Chang B, Yu T, Han A. MEMS micro-coils for magnetic neurostimulation. Biosens Bioelectron. 2023 May 1;227:115143. doi: 10.1016/j.bios.2023.115143.
- Krogsgaard A, Sperling L, Dahlqvist M, Thomsen K, Vydmantaite G, Li F, Thunemann M, Lauritzen M, Lind BL. PV interneurons evoke astrocytic Ca2+responses in awake mice, which contributes to neurovascular coupling. Glia 2023 Mar 30. doi: 10.1002/glia.24370.
- Bakhtiari A, Benedek K, Law I, Fagerlund B, Mortensen EL, Osler M, Lauritzen M, Larsson HBW, Vestergaard MB. Early cerebral amyloid-β accumulation and hypermetabolism are associated with subtle cognitive deficits before accelerated cerebral atrophy. G 2024 Feb;46(1):769-782. doi: 10.1007/s11357-023-01031-w.
- Zhukov O, He C, Soylu-Kucharz R, Cai C, Lauritzen AD, Aldana BI, Björkqvist M, Lauritzen M, Kucharz K. Preserved blood-brain barrier and neurovascular coupling in female 5xFAD model of Alzheimer's disease. Front Aging Neurosci. 2023 May 5;15:1089005. doi: 10.3389/fnagi.2023.1089005.
- Gollion C, Christensen RH, Ashina H, Al-Khazali HM, Fisher PM, Amin FM, Lauritzen M, Ashina M. Somatosensory migraine auras evoked by bihemispheric cortical spreading depression events in human parietal cortex. J Cereb Blood Flow Metab. 2024 Oct 13:271678X241290606. doi: 10.1177/0271678X241290606.
- Kirkegaard JB, Kutuzov NP, Netterstrøm R, Darkner S, Lauritzen M, Lauze F. (2024). In Vivo Deep Learning Estimation of Diffusion Coefficients of Nanoparticles. In: Linguraru, M.G., et al.Medical Image Computing and Computer Assisted Intervention – MICCAI 2024. MICCAI 2024. Lecture Notes in Computer Science, vol 15002. Springer, Cham. https://doi.org/10.1007/978-3-031-72069-7_20
- Vestergaard MB, Bakhtiari A, Osler M, Mortensen EL, Lindberg U, Law I, Lauritzen M, Benedek K, Larsson HBW. The cerebral blood flow response to neuroactivation is reduced in cognitively normal men with β-amyloid accumulation. Alzheimers Res Ther. 2025 Jan 4;17(1):4. doi: 10.1186/s13195-024-01652-z.
- Ghazi MM, Urdanibia-Centelles O, Bakhtiari A, Fagerlund B, Vestergaard MB, Larsson HBW, Mortensen EL, Osler M, Nielsen M, Benedek K, Lauritzen M. Cognitive aging and reserve factors in the Metropolit 1953 Danish male cohort. 2024 Nov 21. doi: 10.1007/s11357-024-01427-2.
- Mao X, Shenton N, Puthusserypady S, Lauritzen MJ, Benedek K. Auditory steady state response can predict declining EF in healthy elderly individuals. Frontiers in Aging Neuroscience 17, Volume 17 - 2025 | https://doi.org/10.3389/fnagi.2025.1516932
- Zhang X, Tao L, Nygaard AH, Dong Y, Groves T, Hong X, Goddard CM, He C, Postnov D, Allodi I, Lauritzen MJ, Cai C. Aging alters calcium signaling in vascular mural cells and drives remodeling of neurovascular coupling in the awake brain. .J Cereb Blood Flow Metab. 2025 Feb 13:271678X251320455. doi: 10.1177/0271678X251320455.

Lab members
| Name | Title | Job responsibilities | |
|---|---|---|---|
| Search in Name | Search in Title | Search in Job responsibilities | |
| Aleksandra Petrovskaia | PhD Fellow | Lauritzen Lab |
|
| Chen He | Postdoc | Lauritzen Lab |
|
| Joshua James Shrouder | Postdoc | Lauritzen Lab |
|
| Krzysztof Kucharz | Associate Professor | Lauritzen lab |
|
| Martin Lauritzen | Professor | Lauritzen lab |
|
| Nadia Østergaard Jørgensen | Laboratory Assistant | Lauritzen Lab |
|
| Nikolay Kutuzov | Assistant Professor | Lauritzen Lab |
|
| Xiao Zhang | Assistant Professor | Cai Lab, Lauritzen Lab |
|
